[ad_1]
The ocean floor as we know it is dissolving rapidly as a result of human activity.
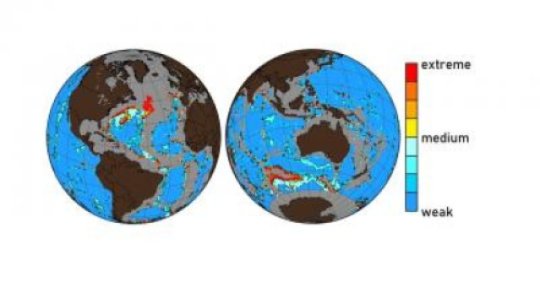
Normally the deep sea bottom is a chalky white. It’s composed, to a large extent, of the mineral calcite (CaCO3) formed from the skeletons and shells of many planktonic organisms and corals. The seafloor plays a crucial role in controlling the degree of ocean acidification. The dissolution of calcite neutralizes the acidity of the CO2, and in the process prevents seawater from becoming too acidic. But these days, at least in certain hotspots such as the Northern Atlantic and the southern Oceans, the ocean’s chalky bed is becoming more of a murky brown. As a result of human activities the level of CO2 in the water is so high, and the water is so acidic, that the calcite is simply being dissolved.
The McGill-led research team who published their results this week in a study in PNAS believe that what they are seeing today is only a foretaste of the way that the ocean floor will most likely be affected in future.
Long-lasting repercussions
“Because it takes decades or even centuries for CO2 to drop down to the bottom of the ocean, almost all the CO2 created through human activity is still at the surface. But in the future, it will invade the deep-ocean, spread above the ocean floor and cause even more calcite particles at the seafloor to dissolve,” says lead author Olivier Sulpis who is working on his PhD in McGill’s Dept. of Earth and Planetary Sciences. “The rate at which CO2 is currently being emitted into the atmosphere is exceptionally high in Earth’s history, faster than at any period since at least the extinction of the dinosaurs. And at a much faster rate than the natural mechanisms in the ocean can deal with, so it raises worries about the levels of ocean acidification in future.”
In future work, the researchers plan to look at how this deep ocean bed dissolution is likely to evolve over the coming centuries, under various potential future CO2 emission scenarios. They believe that it is critical for scientists and policy makers to develop accurate estimates of how marine ecosystems will be affected, over the long-term, by acidification caused by humans.
How the work was done
Because it is difficult and expensive to obtain measurements in the deep-sea, the researchers created a set of seafloor-like microenvironments in the laboratory, reproducing abyssal bottom currents, seawater temperature and chemistry as well as sediment compositions. These experiments helped them to understand what controls the dissolution of calcite in marine sediments and allowed them to quantify precisely its dissolution rate as a function of various environmental variables. By comparing pre-industrial and modern seafloor dissolution rates, they were able to extract the anthropogenic fraction of the total dissolution rates.
The speed estimates for ocean-bottom currents came from a high-resolution ocean model developed by University of Michigan physical oceanographer Brian Arbic and a former postdoctoral fellow in his laboratory, David Trossman, who is now a research associate at the University of Texas-Austin.
“When David and I developed these simulations, applications to the dissolution of geological material at the bottom of the oceans were far from our minds. It just goes to show you that scientific research can sometimes take unexpected detours and pay unexpected dividends,” said Arbic, an associate professor in the University of Michigan Department of Earth and Environmental Sciences.
Trossman adds: “Just as climate change isn’t just about polar bears, ocean acidification isn’t just about coral reefs. Our study shows that the effects of human activities have become evident all the way down to the seafloor in many regions, and the resulting increased acidification in these regions may impact our ability to understand Earth’s climate history.”
“This study shows that human activities are dissolving the geological record at the bottom of the ocean,” says Arbic. “This is important because the geological record provides evidence for natural and anthropogenic changes.”
Story Source:
Materials provided by McGill University. Note: Content may be edited for style and length.
[ad_2]